Presentation
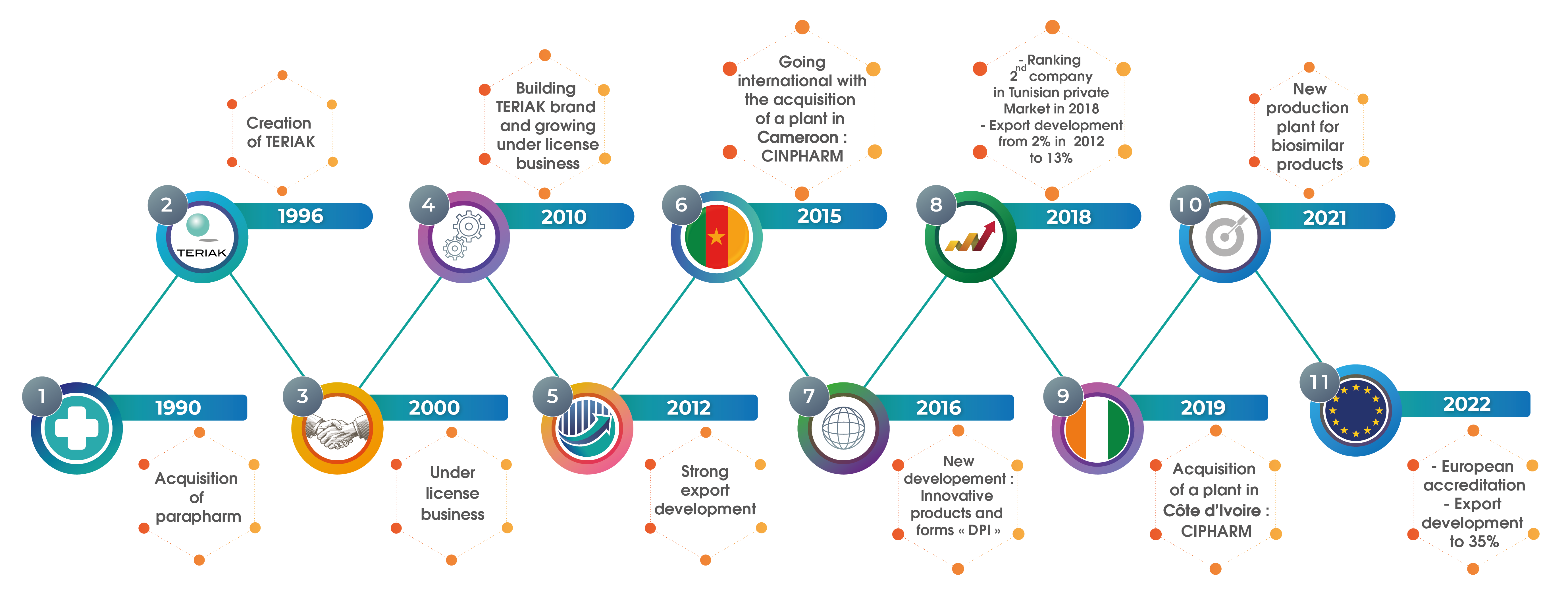
TERIAK Laboratories, a subsidiary of KILANI group is a major player in the Tunisian pharmaceutical industry. Since the establishment of the company in 1996, we have built strong partnerships with leading multinational groups. Our development has been through ethics and values shared with our500 employees.
TERIAK has three production sites. Sites were entirely designed in accordance with GMP standards and are regularly audited by the licensors and health authorities.
- Jebel Ouest Site (Tunisia) : Forms tablets, coated tablets, sachets of powder, liquid sachets, capsules, dry syrups, syrups, pasty forms, suppositories, suspensions.
- El Fejja Site (Tunisia): Inhalation capsules, sterile forms.
- Cinpharm Site (Cameroon, Douala): liquid forms; tablets, coated tablets, powder sachets, liquid sachets, capsules, dry syrups, capsules; as well as B-lactams products.
- Abidjan Site (Côte d'Ivoire) : Capsules, tablets, syrups.
TERIAK manufactures under license for leading pharmaceutical multinational companies (five of these licensors are ranked among the top eight pharmaceutical companies in the world).
TERIAK develops also its own high quality generic products.
TERIAK has developed particular synergies with other subsidiaries of KILANI Group to control distribution and medical promotion.
With its commitment to professional ethics, strong involvement of all staff and putting quality at the heart of its business, TERIAK is now the second local manufacturer.
History